Tirzepatide has emerged as a significant area of interest within the fields of endocrinology and metabolic health. This synthetic peptide, a novel pharmaceutical agent, has garnered substantial attention for its dual-action mechanism targeting both glycemic control and weight management. The growing public and scientific curiosity surrounding tirzepatide is evident in global search trends, particularly in Western countries grappling with high prevalences of obesity and type 2 diabetes. Google Trends data illustrates an exponential rise in search interest, reflecting increased accessibility and a heightened awareness of its potential applications. This article aims to provide a comprehensive, science-backed overview of tirzepatide, exploring its mechanism of action, clinical findings, therapeutic applications, and safety considerations.
Mechanism of Action
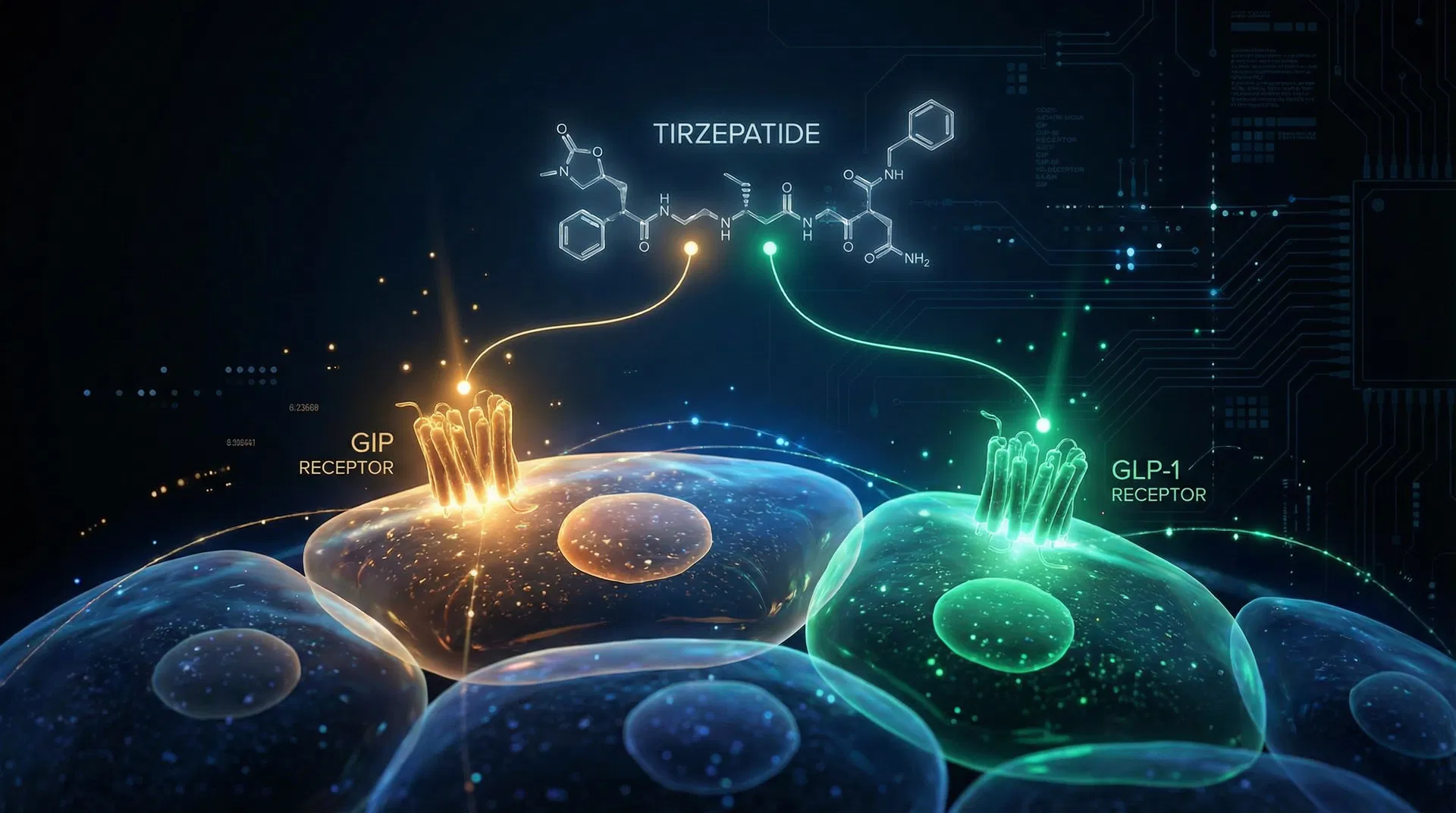
Tirzepatide operates as a dual agonist for two key incretin receptors: the glucagon-like peptide-1 (GLP-1) receptor and the glucose-dependent insulinotropic polypeptide (GIP) receptor Farzam et al., 2024. Incretins are gut hormones released after nutrient intake, playing a crucial role in glucose homeostasis.
The activation of these receptors by tirzepatide leads to a multifaceted physiological response:
- Stimulation of Insulin Release: When blood glucose levels are elevated, tirzepatide enhances the glucose-dependent secretion of insulin from pancreatic beta cells. This mechanism helps to lower postprandial and fasting glucose levels without significantly increasing the risk of hypoglycemia in euglycemic states Farzam et al., 2024.
- Reduction of Glucagon Secretion: Tirzepatide also suppresses the secretion of glucagon, a hormone that raises blood glucose levels by promoting hepatic glucose production. This dual action on insulin and glucagon contributes significantly to improved glycemic control.
- Delayed Gastric Emptying: Similar to GLP-1 receptor agonists, tirzepatide slows down gastric emptying. This contributes to a feeling of fullness and satiety, which can lead to reduced food intake and subsequent weight loss.
- Increased Satiety and Reduced Appetite: By influencing central nervous system pathways involved in appetite regulation, tirzepatide helps to reduce hunger and increase feelings of satiety, further supporting its weight-reducing effects.
- Adiponectin Level Modulation: Research indicates that tirzepatide can increase levels of adiponectin, an adipokine that plays a role in glucose and lipid metabolism and has anti-inflammatory properties. Higher adiponectin levels are generally associated with improved insulin sensitivity and reduced risk of metabolic diseases.
This synergistic dual agonism of both GLP-1 and GIP receptors is believed to be a key factor in tirzepatide's superior efficacy in both glycemic control and weight reduction compared to GLP-1 receptor agonists alone Farzam et al., 2024.
Clinical Evidence & Research Findings
Extensive clinical trials have evaluated the efficacy and safety of tirzepatide across various populations. The SURPASS clinical trial program has been instrumental in demonstrating its robust effects on glycemic control and body weight.
-
Glycemic Control: Studies have consistently shown that tirzepatide leads to substantial reductions in HbA1c (glycated hemoglobin), a key marker of long-term blood glucose control. For instance, in trials comparing tirzepatide to other antidiabetic medications, tirzepatide demonstrated superior HbA1c reductions, often achieving target glycemic goals in a higher percentage of patients Farzam et al., 2024. These improvements are attributed to its potent effects on insulin secretion, glucagon suppression, and glucose-dependent mechanisms.
-
Weight Loss: Beyond its glycemic benefits, tirzepatide has shown remarkable efficacy in promoting significant weight loss. In several studies, participants receiving tirzepatide experienced dose-dependent body weight reductions that were often more pronounced than those observed with GLP-1 receptor agonists or insulin Farzam et al., 2024. This weight loss is a critical benefit, particularly given the strong association between obesity and type 2 diabetes and its complications. The mechanisms contributing to weight loss include delayed gastric emptying, increased satiety, and reduced appetite.
-
Cardiovascular Outcomes: While dedicated cardiovascular outcome trials are ongoing, preliminary data and mechanistic insights suggest potential cardiovascular benefits. Improved glycemic control, significant weight loss, and favorable effects on lipid profiles (e.g., reductions in triglycerides and increases in high-density lipoprotein cholesterol) are all factors that could contribute to reduced cardiovascular risk in the long term.
Looking for a physician-guided approach? Telegenix connects you with licensed providers who specialize in peptide and testosterone optimization. Every protocol is built on your bloodwork. Learn more and book a consultation.
Therapeutic Applications
Tirzepatide is currently approved for specific therapeutic indications, with ongoing research exploring its potential in other areas.
-
Type 2 Diabetes Mellitus: The primary approved indication for tirzepatide is for the treatment of adults with type 2 diabetes mellitus. It is used as an adjunct to diet and exercise to improve glycemic control. Its dual agonism provides a powerful tool for managing hyperglycemia while also addressing the common comorbidity of obesity in this patient population Farzam et al., 2024.
-
Obesity Treatment (Off-Label Use and Research): While not yet formally approved for obesity treatment in all regions, tirzepatide is increasingly being used off-label for this purpose, particularly in individuals with a high body mass index (BMI) who may or may not have type 2 diabetes. The substantial weight loss observed in clinical trials, often exceeding 15-20% of baseline body weight, underscores its potential as a highly effective anti-obesity medication Farzam et al., 2024. Research is actively exploring its role in broader obesity management.
-
Nonalcoholic Fatty Liver Disease (NAFLD): There is emerging interest in tirzepatide's potential for treating nonalcoholic fatty liver disease (NAFLD), including its more severe form, nonalcoholic steatohepatitis (NASH). Given that NAFLD is closely linked to insulin resistance, obesity, and type 2 diabetes, the improvements in glycemic control, weight loss, and potentially direct effects on hepatic fat accumulation make tirzepatide a promising candidate for further investigation in this area. Reductions in liver fat content have been observed in studies, suggesting a beneficial impact on liver health.
Safety Profile & Side Effects
While tirzepatide offers significant therapeutic benefits, it is associated with a distinct safety profile and potential side effects that warrant careful consideration. Monitoring and management of these adverse reactions are crucial for patient well-being Liu et al., 2024.
The most common side effects are typically gastrointestinal in nature and tend to be dose-dependent, often more pronounced during dose escalation:
- Nausea: This is one of the most frequently reported side effects.
- Diarrhea: Another common gastrointestinal complaint.
- Vomiting: Can occur, especially at higher doses or during initial treatment.
- Decreased Appetite: While often a desired effect for weight loss, excessive appetite suppression can lead to inadequate nutrient intake.
These gastrointestinal symptoms often subside over time as the body adapts to the medication. Gradual dose escalation is a strategy employed to mitigate these effects.
Other known risks and potential adverse events include:
- Sinus Tachycardia: An increase in heart rate has been observed in some individuals.
- Acute Kidney Injury: This can occur, primarily secondary to dehydration resulting from severe gastrointestinal side effects such as vomiting and diarrhea. Maintaining adequate hydration is important.
- Hypersensitivity Reactions: Allergic reactions, though rare, can occur.
- Pancreatitis: While rare, there have been reports of acute pancreatitis in patients treated with incretin mimetics, including tirzepatide. Patients should be advised to seek medical attention if they experience severe, persistent abdominal pain.
- Worsening of Diabetic Retinopathy: In some studies, there have been observations of a transient worsening of diabetic retinopathy, particularly in patients with a history of retinopathy and rapid improvements in glycemic control. Regular ophthalmologic examinations are important for individuals with diabetes.
- Cholelithiasis and Cholecystitis: Rapid weight loss, as can be achieved with tirzepatide, is a known risk factor for gallstone formation (cholelithiasis) and subsequent inflammation of the gallbladder (cholecystitis).
- Thyroid C-cell Tumors: Tirzepatide, like other GLP-1 receptor agonists, has been shown to cause dose-dependent and treatment-duration-dependent thyroid C-cell tumors at clinically relevant exposures in rodents. It is contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN-2). The relevance of these findings to humans is currently unknown, but caution is advised Farzam et al., 2024.
Recent studies have also aimed to provide new perspectives on monitoring and managing adverse drug reactions, identifying potential novel and unexpected adverse event signals associated with tirzepatide use Liu et al., 2024, Li et al., 2025. While tirzepatide was associated with an increased risk of specific adverse events, its overall safety profile has been noted to be similar to that of other GLP-1 receptor agonists Caruso et al., 2024.
Dosing Considerations
Tirzepatide is administered via subcutaneous injection once weekly. Research protocols typically involve a gradual dose escalation to improve tolerability and minimize gastrointestinal side effects.
- Initial Dosing: Treatment usually begins with a lower starting dose, such as 2.5 mg once weekly.
- Dose Escalation: The dose is then typically increased incrementally (e.g., by 2.5 mg) every four weeks until a maintenance dose is reached. Common maintenance doses used in clinical trials range from 5 mg, 10 mg, to 15 mg once weekly.
- Maximum Dose: The maximum recommended dose in clinical studies is typically 15 mg once weekly.
The specific dosing regimen and titration schedule are determined by the treating healthcare provider based on individual patient response, glycemic control, weight loss goals, and tolerability. It is important to note that these are general protocols observed in research and do not constitute specific recommendations.
Key Takeaways
- Tirzepatide is a dual GLP-1 and GIP receptor agonist that significantly improves glycemic control and promotes substantial weight loss in adults with type 2 diabetes Farzam et al., 2024.
- Its mechanism involves stimulating glucose-dependent insulin release, suppressing glucagon, delaying gastric emptying, and enhancing satiety Farzam et al., 2024.
- Primary benefits include improved HbA1c, significant body weight reduction, and potential applications in conditions like nonalcoholic fatty liver disease.
- Common side effects are primarily gastrointestinal, such as nausea, diarrhea, and vomiting, which often improve with continued use and dose titration Liu et al., 2024.
- Important contraindications include a personal or family history of medullary thyroid carcinoma or MEN-2 due to observed C-cell tumors in rodents Farzam et al., 2024.
Related Comparison: Ozempic vs Mounjaro: Complete Comparison [blocked]
References
Disclaimer: This article is intended for educational purposes only and should not be considered medical advice. It is crucial to consult with a qualified healthcare professional for any health concerns or before making any decisions related to your health or treatment.
Ready to put this research into practice? Telegenix provides evidence-based peptide and TRT [blocked] protocols supervised by licensed providers. Get a personalized plan built around your bloodwork. Start with a free consultation.
Related Reading
Explore more in-depth guides on related topics:
- Semaglutide: What the Science Actually Says — A PubMed-Backed Review [blocked]
- Retatrutide: What the Science Actually Says — A PubMed-Backed Review [blocked]
- Tirzepatide: The Dual-Action Peptide for Diabetes and Weight Loss [blocked]
- Cagrilintide: What the Science Actually Says — A PubMed-Backed Review [blocked]
- Retatrutide: The Triple-Agonist Peptide Revolutionizing Obesity Treatment [blocked]
For a comprehensive overview, see our Complete Guide to Peptide Therapy [blocked].