Compounded semaglutide vs Brand Ozempic: Cost, Safety, and Efficacy in 2026
1. Introduction
The landscape of metabolic health and weight management has been dramatically reshaped by the advent of GLP-1 weight loss guide receptor agonists like semaglutide. For millions, these medications offer unprecedented efficacy in managing type 2 diabetes and achieving significant best peptides for weight loss. However, the high cost of brand-name products such as Ozempic and Wegovy, often exceeding $1,000 to $1,500 per month without insurance, has created a significant barrier to access.
This financial hurdle led to a surge in the use of compounded semaglutide, which, until recently, faced regulatory uncertainty. The FDA's landmark decision in March 2026 to allow compounding pharmacies to resume producing semaglutide has fundamentally transformed the market. With compounded versions available for a fraction of the cost, typically $150-$400 per month, patients are now faced with a critical choice. This comprehensive guide aims to break down the differences between brand-name and compounded semaglutide, examining cost, safety, and efficacy to help patients make an informed decision in 2026.
2. What Is Compounded Semaglutide?
Compounding is the process by which a licensed pharmacist or physician combines, mixes, or alters ingredients to create a medication tailored to the needs of an individual patient. This is distinct from manufacturing, where drugs are produced in large quantities by pharmaceutical companies.
Compounded semaglutide refers to a version of the drug prepared by a compounding pharmacy. These pharmacies operate under two main classifications:
- 503A Compounding Pharmacies: These are traditional state-licensed pharmacies that compound medications based on individual patient prescriptions. They must adhere to state pharmacy board regulations and the United States Pharmacopeia (USP) standards for quality.
- 503B Outsourcing Facilities: These are federally registered facilities that are permitted to compound sterile drugs in bulk without patient-specific prescriptions, often in anticipation of future demand. They are subject to more stringent FDA oversight, including current Good Manufacturing Practice (cGMP) requirements, similar to pharmaceutical manufacturers, but for compounded products.
The legal framework for compounded semaglutide was significantly clarified by the FDA's 2026 reclassification. While compounded semaglutide uses the same active pharmaceutical ingredient (API), semaglutide, it may differ from brand-name products in its formulation, concentration, and inactive ingredients (excipients). For example, compounded versions might be formulated as a sterile injectable solution, but the specific buffers, preservatives, or other excipients used can vary.
3. The FDA Reclassification Timeline
The regulatory journey of compounded semaglutide has been complex and dynamic:
- March 2024: The FDA officially added semaglutide to its Drug Shortage List. This designation is crucial because under Section 503A and 503B of the Federal Food, Drug, and Cosmetic Act, compounding pharmacies are permitted to compound drugs that are on the FDA's shortage list, even if those drugs are otherwise protected by patents.
- 2024-2025: During this period, compounding pharmacies legally produced and dispensed semaglutide, providing a vital alternative for patients unable to access or afford brand-name versions. This period saw a significant expansion of compounded GLP-1 access.
- Late 2025: As manufacturing capacity for brand-name Ozempic and Wegovy increased, the FDA signaled its intention to remove semaglutide from the shortage list. This move would have effectively ended the legal compounding of semaglutide, sparking widespread concern among patients and providers.
- March 2026: In a significant reversal, influenced by public outcry, advocacy efforts, and potentially political pressure (including from figures like RFK Jr. who championed broader access to compounded medications), the FDA announced a new policy. This policy reclassified several peptides, including semaglutide, to allow for continued compounding, even if the brand-name product was no longer on the official shortage list. This decision recognized the ongoing access and affordability issues faced by many patients. The FDA's specific guidance documents in early 2026 outlined the conditions under which compounding of these peptides would be permissible, focusing on patient need and quality assurance.
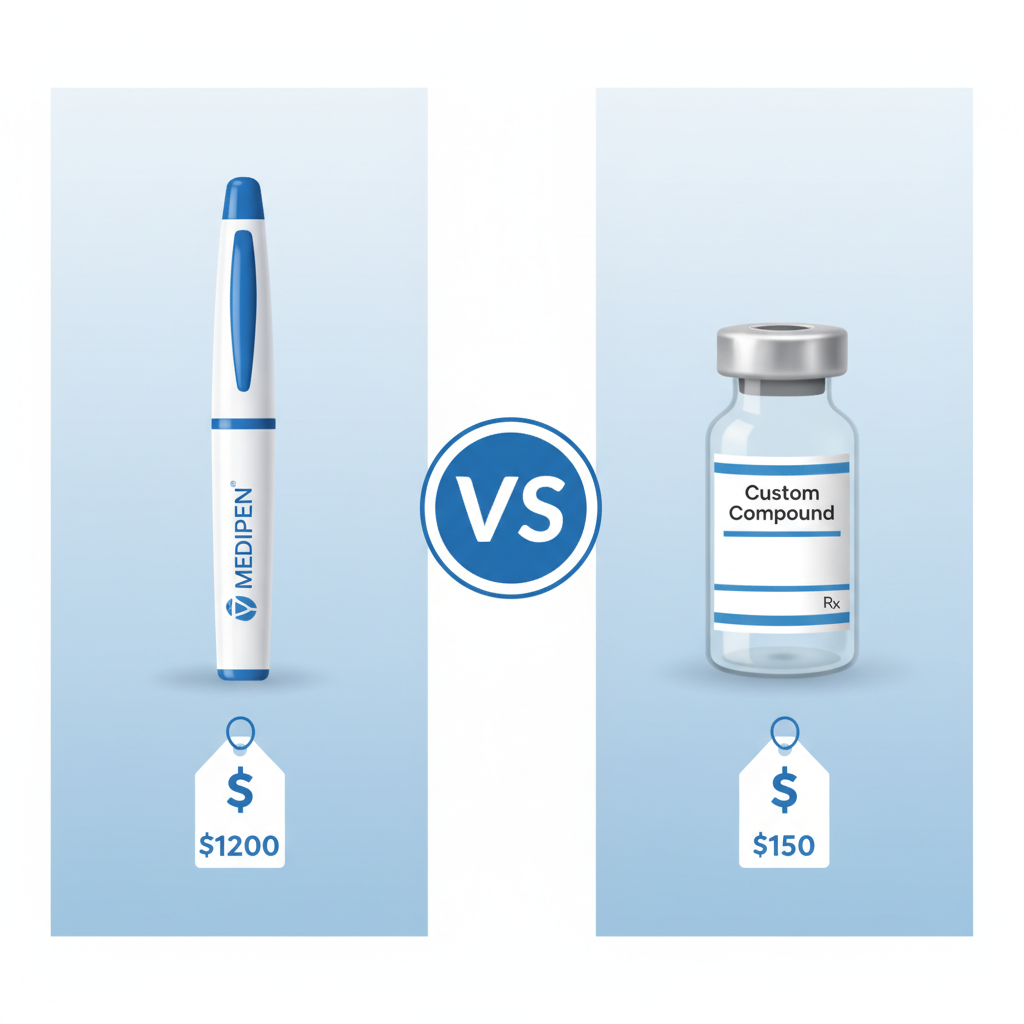
4. Cost Comparison: Brand vs Compounded
The cost disparity is arguably the most significant factor driving patient choice.
| Product | Typical Monthly Cost (without insurance) |
|---|---|
| Brand Ozempic (0.25mg-1mg) | $800 - $1,500 |
| Brand Wegovy (2.4mg) | $1,300 - $1,700 |
| Compounded Semaglutide | $150 - $400 (depending on dose, pharmacy, and excipients) |
Insurance Coverage Landscape in 2026: Insurance coverage for brand-name Ozempic (for type 2 diabetes) and Wegovy (for weight management) remains highly variable. Many plans require prior authorization, step therapy, or may not cover weight loss medications at all. Even with coverage, high deductibles and co-pays can leave patients with substantial out-of-pocket expenses.
Compounded semaglutide is generally not covered by insurance. This is because compounded drugs are not FDA-approved in the same way manufactured drugs are, and insurers typically only cover FDA-approved products. However, the lower cash price of compounded versions often makes them more affordable than brand-name drugs, even for patients with insurance coverage that doesn't fully cover the brand.
5. Efficacy: Is Compounded Semaglutide as Effective?
The fundamental premise of compounded semaglutide's efficacy rests on the fact that it contains the identical active pharmaceutical ingredient (API), semaglutide. Semaglutide is a GLP-1 receptor agonist that mimics the effects of naturally occurring GLP-1, leading to improved glycemic control and reduced appetite.
The efficacy of semaglutide was robustly demonstrated in clinical trials. For instance, the STEP 1 trial (Wilding et al., 2021, PMID: 33567185) showed that participants receiving 2.4 mg semaglutide once weekly achieved an average body weight loss of 14.9% from baseline over 68 weeks, compared to 2.4% in the placebo group. This translates to an average weight loss of 15-17% of body weight, a level of efficacy previously unseen with non-surgical interventions.
Bioequivalence Concerns: While the API is the same, concerns about bioequivalence can arise. Bioequivalence refers to the rate and extent to which the active ingredient is absorbed into the bloodstream and becomes available at the site of drug action. Differences in formulation (e.g., excipients, pH, sterility) could theoretically impact absorption, stability, or patient response.
Importance of Third-Party Testing (Certificate of Analysis - COA): To mitigate these concerns, reputable compounding pharmacies will provide a Certificate of Analysis (COA) for their compounded products. A COA should confirm:
- Potency: That the medication contains the stated amount of active ingredient.
- Purity: That it is free from contaminants.
- Sterility: For injectable products, that it is free from microbial contamination.
Real-world Outcomes: Anecdotal evidence and growing clinical experience suggest that properly compounded semaglutide, when sourced from reputable pharmacies, can produce similar efficacy to brand-name versions. Patients often report comparable weight loss and glycemic control. However, it is crucial to understand that these real-world observations lack the rigorous, controlled, large-scale clinical trial data that underpin the efficacy claims of brand-name drugs.
6. Safety Considerations
Safety is paramount, and this is where the differences between FDA-approved manufacturing and compounding pharmacies are most pronounced.
Quality Control Differences:
- FDA-Approved Manufacturing: Brand-name drugs undergo a rigorous FDA approval process, including extensive clinical trials, strict manufacturing standards (cGMP), and continuous post-market surveillance. Every batch is subject to stringent quality control, ensuring consistent purity, potency, and sterility.
- Compounding Pharmacies: While compounding pharmacies are regulated, the oversight is generally less centralized and comprehensive than for pharmaceutical manufacturers. 503A pharmacies are regulated by state boards of pharmacy, which vary in their stringency. 503B outsourcing facilities face more federal FDA oversight, including cGMP requirements, which offers a higher level of assurance.
Sterility Testing and Potency Verification: For injectable semaglutide, sterility testing is critical to prevent infections. Reputable compounding pharmacies should conduct and provide documentation of sterility testing for every batch of injectable products. Similarly, potency verification ensures that the patient receives the correct dose, preventing both underdosing (ineffective) and overdosing (increased side effects).
FDA's Concerns about Compounded GLP-1s: The FDA has historically expressed concerns about compounded GLP-1s, primarily revolving around:
- Lack of FDA approval: Compounded drugs are not individually evaluated or approved by the FDA for safety and efficacy.
- Quality variability: The potential for inconsistent quality, potency, and